The data-heavy medical field has long been seen as fertile ground for artificial intelligence (AI), where machine learning and deep learning techniques could crunch through mountains of data to drive everything from research to personalized medicine. Here at The Next Platform, we’ve talked about using pattern recognition, predictive analytics and AI to identify health risks, and in a poll at the World Medical Innovation Forum earlier this year, 98 percent of respondents said healthcare will be become better or much better through AI.
The enthusiasm level around AI in healthcare is high, but its practical use up to this point has been fairly limited, according to Kimberly Powell, vice president of healthcare at Nvidia. While the promise of driving preventative healthcare and personalized medical plans is out there, much of that promise is as yet unfulfilled.
There has “been a really remarkable record of AI in medicine, but has been largely focused in and around medical imaging,” Powell said. “As you see today, there are dozens of algorithms making their way through FDA approval. There are several dozen more that have been implemented to transform the practice of radiology, like triaging patients or auto-protocoling machines and so forth. But we haven’t really tapped into what I call a lot of the other human healthcare data. Those are things like genomics, your DNA profile, your health records with imaging, and then all the wearable technology that is becoming very accessible to consumers as well as being implemented in the provider space.”
Nvidia has built much of its business around AI and deep learning in recent years, and has worked with companies like GE to in helping to support the development of cutting-edge medical devices. The GPU-maker is now working with the Scripps Research Translational Institute in hopes of changing that. The two organizations are coming together in a wide-ranging partnership to develop best practices, tools and infrastructure around AI and deep learning to drive the development of AI-based applications that use genomic and digital health sensor data, much of which is available but has largely been untapped. In addition, Scripps and Nvidia will create a center of excellence for AI in genomics and digital sensors.
The aim is to move healthcare forward to where the massive amounts of data are available or can be gathered can be collected, stored and analyzed to accelerate the ability of clinicians to diagnose issues, detect patterns and drive preventative medicine. In a conference call with journalists, Powell compared the situation in healthcare with autonomous driving, noting that with self-driving cars, it’d be better to be able to avoid an accident rather than determine what happened to the car after one.
“If you think about self-driving cars, we use many different sensors – we use cameras, we use Lidar, we use infra-red, we are watching what’s happening to the driver inside the car to understand his surroundings,” she said. “That sensor fusion is allowing us to create a safer driving environment for self-driving cars. That’s exactly the mission here. We’ve been riding on the coattails of the consumer Internet and the real breakthroughs that have happened in AI. They’ve largely been around medical imaging. What needs to happen is we need to develop the very pervasive data set and tools like AI training frameworks that are friendly to genomic and sensor data.”
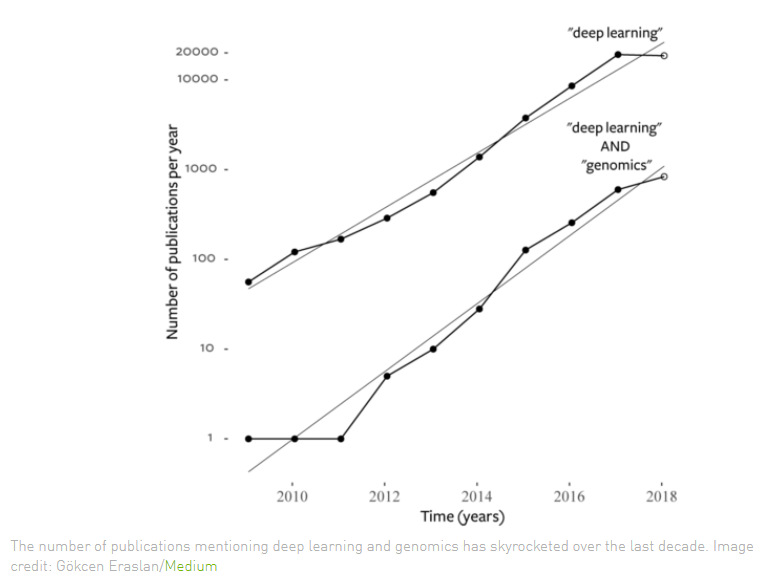
The data is there and it’s growing fast, due to such advances as more affordable genome sequencing equipment and digital health sensors like smartwatches, blood pressure cuffs and glucose monitors. Genomics data is doubling every seven months and the subject of deep learning techniques found in genomic research papers has jumped 40 times in the last four years, according to Nvidia and Scripps. Deep learning approaches can improve the detection of mutations in genomics sequencing and make such work more affordable and accessible.
AI and deep learning will be needed going forward with the amount of data being collected, according to Eric Topol, the Scripps Institute’s founder and director.
“To take all that data together – all the different biologic layers, the digitization with sensors, the imaging and electronic health records – it’s a formidable task that no one yet has achieved,” Topol said. “We are talking about terabytes of data on any individual. We’ve already seen how that can’t be handled by human beings. We need machine support and in order for us to do that, it means bringing the best human minds together to figure out how to get that best machine support.”
Tools will be needed not only for building datasets but also training frameworks that are optimized for sensor and genomics data, which Powell will have to be different from current frameworks that are built for text or images. Tools developed by Nvidia and Scripps also will be made available to outside researchers.
The first area of research in this partnership will be heart rhythm disorders, Topol said. There already is a lot of data available around this, and sensors on wearables like Apple Watches – which now can conduct electrocardiograms – and Fitbits can draw in even more. With such data, the goal is to find markers that can tell researchers and medical professionals what tends to happen before someone is hit with atrial fibrillation, which could help in preventing the condition before it occurs.
“Essentially you can diagnose atrial fibrillation,” he said. “You don’t need deep learning for that. But determining what are the markers – the electrocardiographic markers – that would be precursor to atrial fibrillation, that would be a great step forward because then you could understand at that level – again, adding to genomics and other means – who are the people who could develop this so you could avert that, because here is a risk of stroke with that. It’s ideally something we can prevent and so much of this effort is ultimately here for prevention.”
AI and deep learning methods and algorithms could also be used in analyzing the data that is collected through continuous monitoring devices for such situations as glucose and blood pressure, and they can allow healthcare professionals to better aggregate data from multiple sources to give a more complete picture of a patient’s situation, from how much they sleep, how much physical activity they get and what their diet is like.
Topol noted that there will be FDA-approved continuous blood pressure devices on the market within the next year. The data from that will be important if it can be collected and analyzed.
“We don’t even know what normal blood pressure is in the population,” he said. “We don’t have a clue. The number-one chronic disease of man and we have over 70 million Americans who are affected, and we don’t even know what normal is because we’ve only gotten these one-off values in doctors’ offices or occasionally at home, but now with continuous blood pressure monitoring, people think images have a lot of data? Or a whole genome sequence has a lot of data? They haven’t seen anything yet. Taking blood pressure every minute for a month is a lot of data. How do you analyze that? What does it really mean? And what does that mean when you start studying it in hundreds of thousands of people and collecting it for age and other varying circumstances?”





Be the first to comment